 
The interactive Lattice Energy Calculator provided here makes these calculations more accessible and enhances understanding of this crucial chemical concept. Lattice Energy -436.68-89-(0.5158)-418.8-(-328) kJ/mol -695.48 kJ/mol Lattice Energy -641.8-146-243-(737.7+1450.6)-(2-349) kJ/mol -2521.1 kJ/mol MgO. Calculating lattice energy involves considering the charges of ions and their distance, both of which influence the strength of attractive forces within the lattice. Lattice energy is a fundamental concept in chemistry that impacts the stability and behavior of ionic compounds. This calculator allows you to quickly determine the lattice energy based on the charges of the ions and the distance between them. lattice vibrations by assuming that every atom in a solid oscillates with the. To facilitate the calculation of lattice energy, an interactive Lattice Energy Calculator has been developed. energy and the wave vector ( ka, ky ) is 2 - ( k2 + k2 ). 
The enthalpies of formation of the ionic molecules cannot alone account for this. Ionic solids tend to be very stable compounds. 5.2: Lattice Energy- The Born-Haber cycle is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. 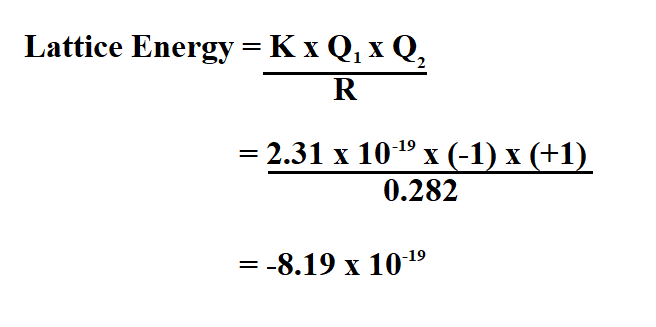
Interactive Lattice Energy Calculator: Making Calculation Easy England: Pearson Education Limited, 2008.174-175. Similarly, as the distance between ions decreases, the lattice energy increases as the ions come closer together, resulting in stronger attractive forces. 1 Q 1 and 2 Q 2 are the charges of the ionsĪs the charges of the ions increase, the lattice energy also increases due to stronger electrostatic attraction.This expression takes into account both the Born interactions as well as the Coulomb attractions. L a tt i ce E n er g y = d k ×∣ Q 1 × Q 2 ∣ The Born-Land equation is a concept originally formulated in 1918 by the scientists Born and Land and is used to calculate the lattice energy (measure of the strength of bonds) of a compound. The formula for calculating lattice energy is as follows: Lattice energy is influenced by two main factors: the charges of the ions involved and the distance between them. Calculating Lattice Energy: The Importance of Ion Charges and Distance This energy is a result of the attractive electrostatic forces between ions of opposite charges and plays a significant role in various chemical processes. It represents the energy released when gaseous ions come together to form a solid ionic lattice. Lattice energy is a crucial concept in the field of chemistry, particularly in understanding the stability and properties of ionic compounds. The Born exponent is typically between 5 and 12.Understanding Lattice Energy and Its Calculation The Born–Landé equation gives an idea to the lattice energy of a system. Heat of formation lattice energy + heat of atomization + dissociation energy + (sum of Ionization energies) + (sum of electron affinities) The Born-Haber Cycle applies Hess’s law to calculate lattice energies by juxtaposing a given ionic compound’s enthalpy change of formation to the enthalpy required to form gaseous ions from its components. According to this equation, stronger interactions occur between ions with larger charges and smaller radii. E = − N A M z + z − e 2 4 π ε 0 r 0 ( 1 − 1 n ) Calculated lattice energies Lattice energy depends on the strength of interactions between cations and anions in the lattice, which we can estimate using Coulomb's law: F (qq)/r. In 1918 Max Born and Alfred Landé proposed that the lattice energy could be derived from the electrostatic potential of the ionic lattice and a repulsive potential energy term. The Born–Landé equation is a means of calculating the lattice energy of a crystalline ionic compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
